Introduction
In circa 1500 Leonardo da Vinci said: "We know more about the movement of celestial bodies than about the soil underfoot". Although our knowledge of both these fields of science has developed immensely over the last five centuries, this statement undoubtedly remains true. Soil life, and especially the soil microbial system, is very dynamic and sensitive, and the potential impacts on its composition are so vast in number that it is very difficult to predict its characteristics under all possible circumstances.
Since the living part of soil consists of a huge number of different species that live in a dynamic ecosystem, where they prey on one another and other times live symbiotically together, the science of soil microbiology is complicated. As a result, very few scientists will claim to be specialists in more than one or two components of soil microbiology. Because of the interest in this topic that has developed among grape producers in recent years, this article attempts to apply relevant elements of this biological science to grape production systems and to address some of the factors that impact vineyard soil-ecosystems.
Soil Organic Matter
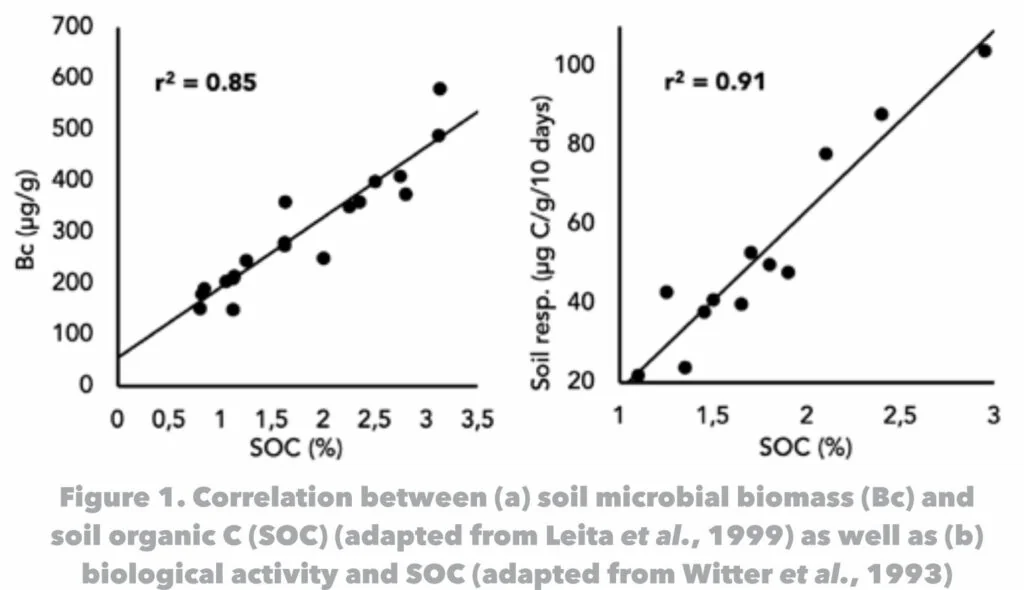
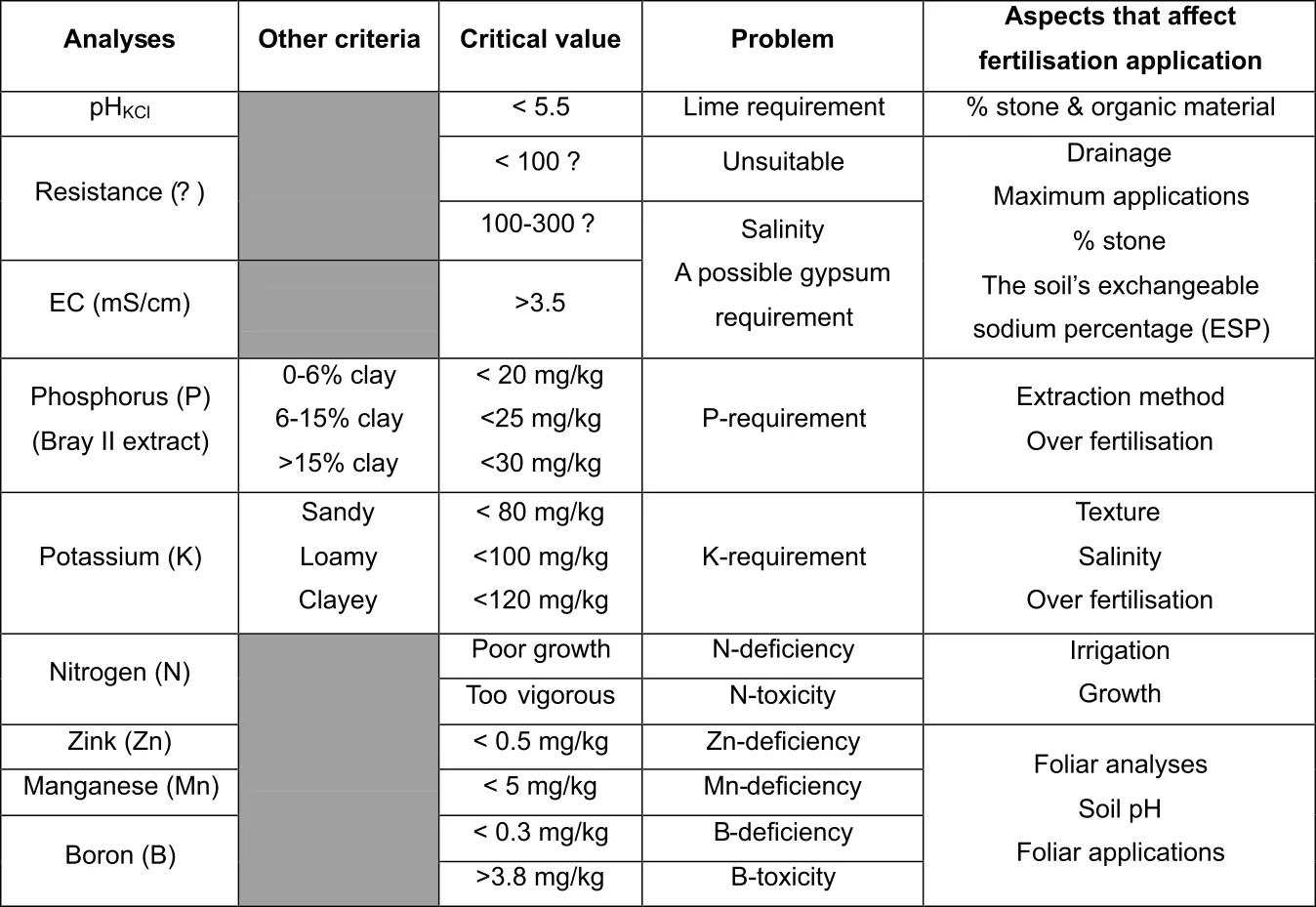
Organic matter is a critical component in the microbial ecosystem of soil since it is the energy source of soil micro-organisms. With a reduction in soil organic matter content, microbial activity decreases. The presence of soil organic matter therefore ensures microbial activity and affects the characteristics of the soil in various ways. Whilst attempting to manage his vineyard soil responsibly, a viticulturalist will benefit from having some knowledge regarding the properties of soil organic matter.
Organic matter is defined as all carbon compounds formed by living organisms. In soil it consists of three components, namely dead organic matter, living parts of plants (eg. roots) and living microbes and animals (Figure 1). Of these three, the first is the largest component, comprising between 70% and 90% of the soil organic matter (Gregorich et al., 1997). Recently died plant material, soil microbes and insects/animals are in a very dynamic state in that it quickly develops into humus. As a result, humus is the largest component of the dead organic matter fraction.
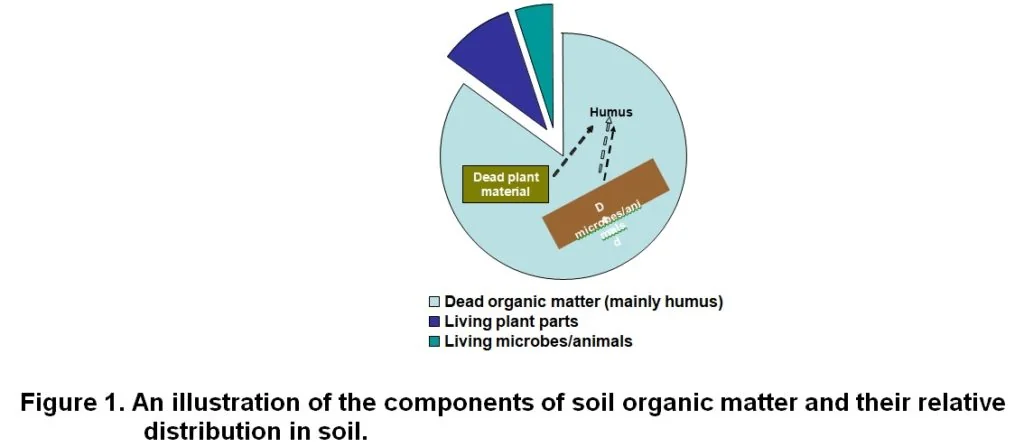
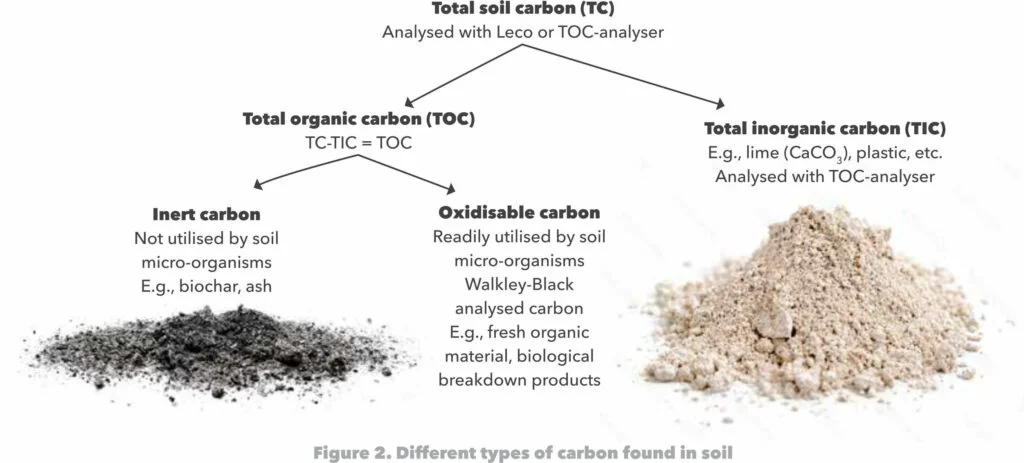
The dead organic matter (humus and fresh soil organic matter) consists of various chemical compounds that individually plays a role in the ecosystem, breaks down at different rates, and gives each soil its own unique character. The most important compounds, that make up the bulk of the material, are: (1) cellulose, (2) lignin, (3) chitin, (4) lipids, (5) protein, and (6) simple soluble molecules.
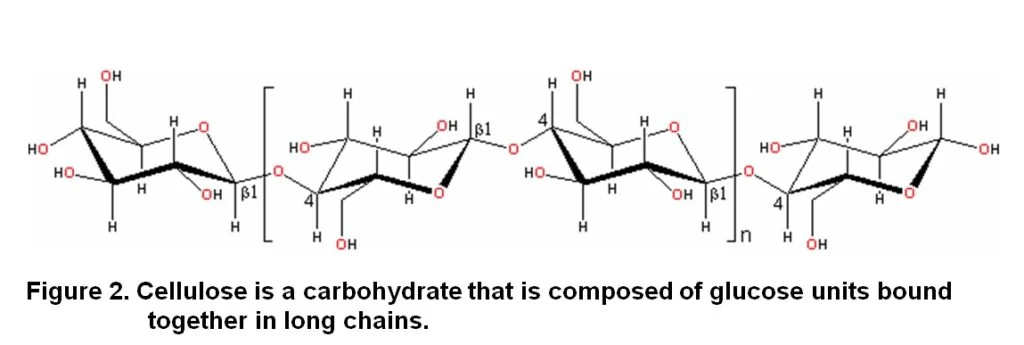
Cellulose is a carbohydrate that composes of glucose that is bound together in long chains (Figure 2). Carbon (C), oxygen (O) and hydrogen (H) are the main elements of this compound. It is prominent in woody substances, straw, stubble and leaves. Through hydrolysis fungi and bacteria convert it to carbon dioxide (CO2) or cell carbon. Cellulose is relatively resistant to microbial attack and the chemical process of hydrolysis. The initial pace of cellulose hydrolysis is therefore slow, often making it the rate limiting reaction in microbial breakdown of soil organic material.
Lignin is a complex long-chain compound (polymer) that is made up of thousands of rings of C atoms joined together in a long chain. The way in which they are linked up varies along the chain. It also contains only C, H and O, but is aromatic compared to cellulose in that it contains phenolic groups. Lignin binds to cellulose fibers to harden and strengthen cell walls of plants. It is plentiful in older higher woody plants, especially in the branches and trunks (Table 1). The most important characteristic of lignin is its high resistance to enzymatic breakdown. The result is that, with the exception of a few specialized fungi and actinomycetes (unicellular filamentous microorganisms that possess properties intermediate between the fungi and the bacteria), soil organisms cannot digest wood. Decomposed soil organic matter will therefore often end up having high lignin contents but little cellulose.

Chitin is also a polymer, analogous in chemical structure to cellulose, but consists of units of a nitrogenous derivative of glucose. It is, therefore a polysaccharide, with amino sugars as its basic structural unit. Like cellulose, chitin contributes strength and protection to organisms. Soil chitin originates from the remains of insect cells and fungi, as well as some actinomycetes and yeasts. It is mainly broken down by actinomycetes, which proliferate in its presence, especially those that excrete anti-fungal substances.
Lipids are substances produced by organisms that are insoluble in water. It includes a lot of compounds like pigments, fats, waxes, phospholipids, sterols, di- & triglycerides, etc. They generally are hydrophobic. Lipids are broken down by specific groups of bacteria that secrete an enzyme (lipase) which hydrolyze it to break down to fatty acids. Last mentioned are then broken down further through oxidation.
Protein is a group of complex organic compounds, mainly one or more chains of amino acids that are linked together by peptide bonds. It therefore contains C, H, O, sulphur (S) and very characteristically, nitrogen (N). They are broken down very easily through hydrolyses of the peptide bonds by enzymes that are excreted by an innumerable amount of heterotrophic soil organisms (organisms which require carbon in organic form). Thereby the amino acids are released which then serve as sources of C and N to the soil organisms. Because of its high N content, protein contributes to the N in soil organic matter and therefore increases the C:N ratio of the soil.
Simple soluble molecules are usually the product of microbial breakdown of organic matter. Examples are, as mentioned above, amino acids, also acids, sugars, alcohols and aromatic groups. These are readily available for uptake or utilization by the soil micro-organisms.
As dead organic matter breaks down, it forms into the most important part of dead soil organic matter, i.e. humus. Last mentioned can be defined as a mixture of colloidal degradation products that accumulates in the soil because it breaks down slower than the products it was formed from. Humus is therefore relatively resistant to microbial breakdown. One of the major substances of humus is residual lignin. Despite its resistance to microbial breakdown, it oxidizes to give humus its black colour. Furthermore, the phenolic groups also contribute to the characteristic smell of humus. The slow process of humus decay happens in two ways, namely oxidation in hot dry climates and microbial breakdown in warm wet climates. A flux in humus decay usually occurs after a dry spell. This is due to the oxidation that takes place in the dry period whereby C is lost from the humus as CO2 and the chemical composition of the resistant compounds in the humus is altered. Afterwards, when the soil is moistened, an explosion in the population of soil organisms occurs due to the sudden increase in digestible compounds and the favourable climatic conditions.
Breakdown of Soil Organic Matter
By now it must be clear that the process of soil organic matter breakdown is complex. It involves different stages of breakdown taking place simultaneously, with different compounds breaking down at different rates and due to the activity of different organisms (Figure 3). The stages of breakdown can be separated into (1) the colonization and physical fragmentation of fresh organic matter, (2) the chemical alteration of the organic matter and, finally, (3) the release of mineral nutrients.
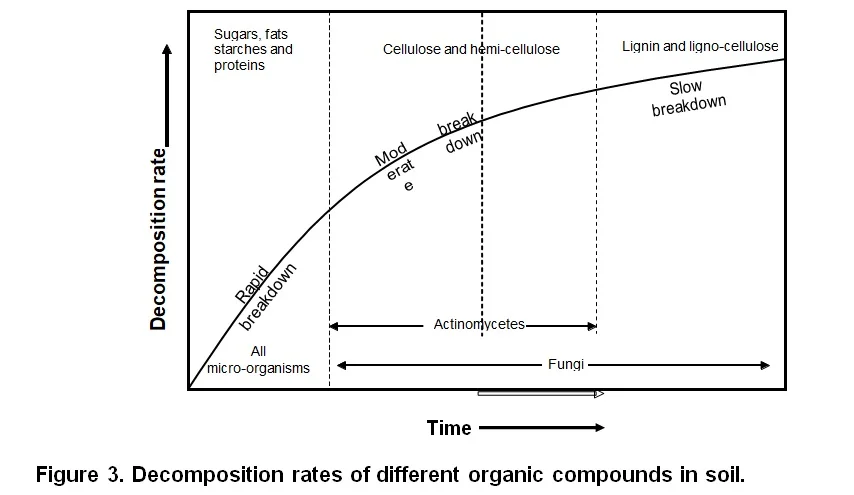
Leaf- and root surfaces are colonised by micro-organisms even before they die. Once the dead organic matter lands on the soil, they immediately excrete enzymes that oxidize the organic matter to obtain energy and C. In the process C is lost through the release of CO2. The result is a decrease in the C:N ratio of the organic matter. Simultaneously, earthworms and other larger soil animals such as mites and ants, start fragmenting the fresh organic matter. The surface area is thereby increased allowing more micro-organisms to colonise and decompose it. Soil animals, especially earthworms, assist with the colonisation process by incorporating organic matter into the soil (where conditions for microbial colonization are more favourable) and mixing it with intestinal microbes as they digest the material. The fungi, bacteria and actinomycetes secrete enzymes to break up the organic compounds which all are, as mentioned above, long and complex chains. Simpler molecules are then formed from which, with further decomposition, are mineral nutrients finally released. This process is called mineralization, i.e. the biological process by which organic compounds are chemically converted to simpler organic compounds and finally to mineral nutrients (Figure 4).

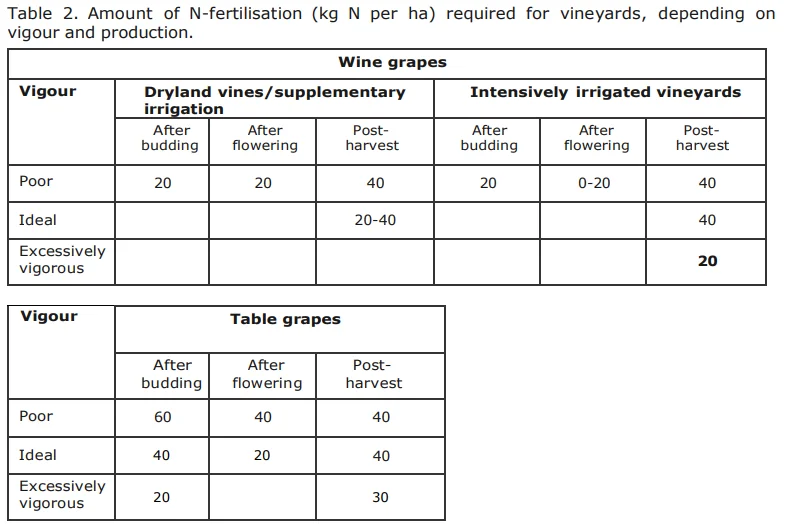
Because of the large amount of N that can be released from incorporated soil organic matter for plant nutrition, N-mineralization is of economic importance. It consists of two steps:
- Ammonification - the release of N as ammonium (NH +) from organic molecules through enzyme activity of bacteria & fungi (Figure 5).
- Nitrification - the release of N as ammonium (NH +) from organic molecules through enzyme activity of bacteria & fungi (Figure 5).
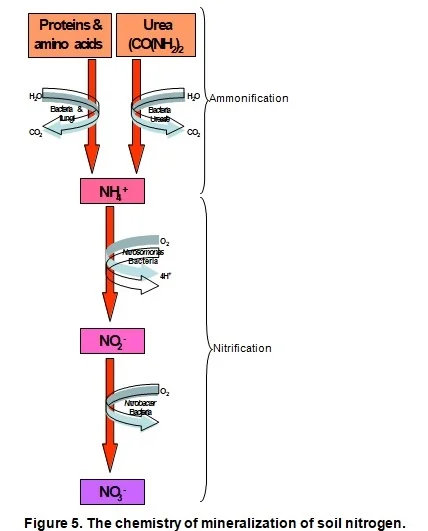
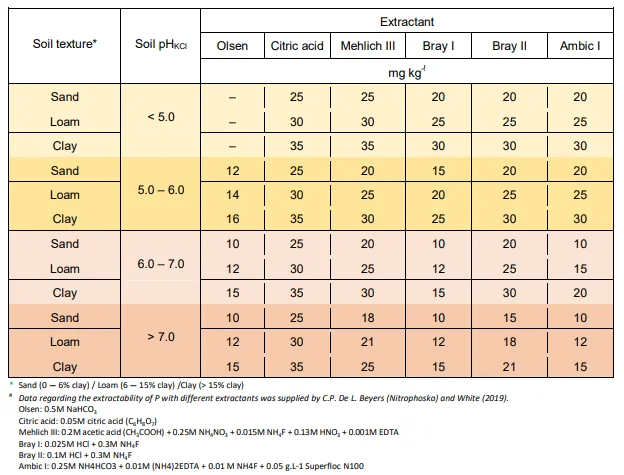
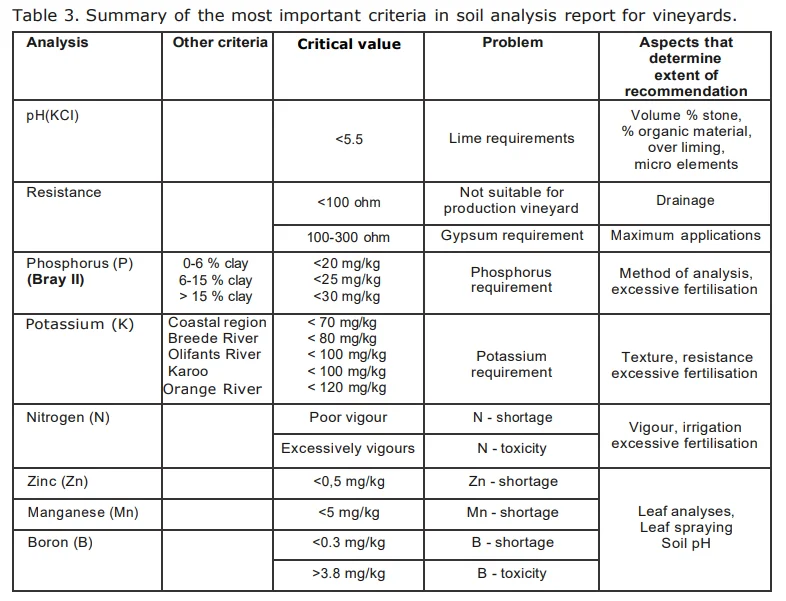
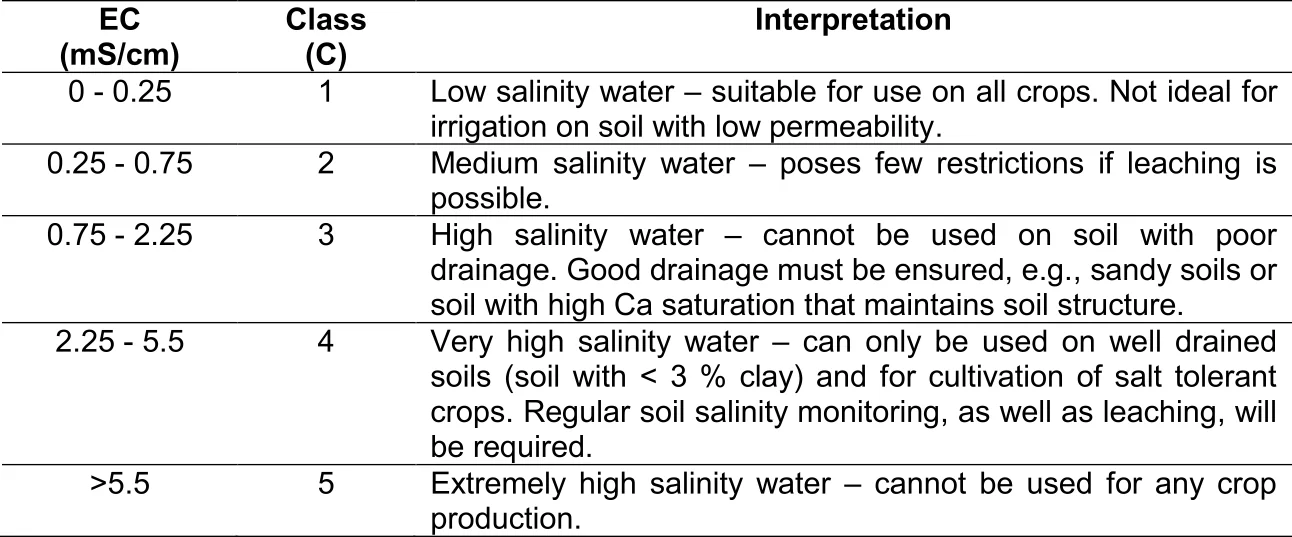
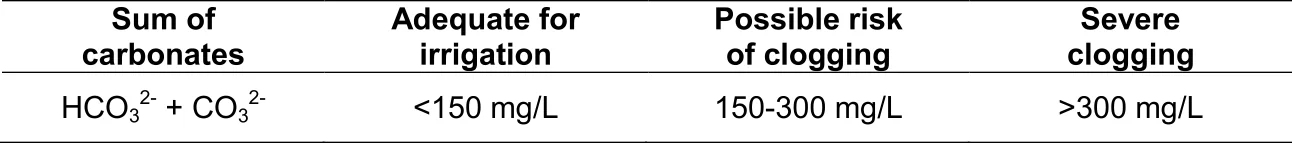
From Figure 5 it can be deduced that urea, being an organic molecule, also requires mineralization before the N therein is released for uptake. This process of urea breakdown, however, is done by a very specific group of bacteria that are able to release an enzyme called "urease". Low microbial activity is the reason why the use of urea is not recommended as fertilizer on coarse sandy soils. Furthermore, it is worthwhile noticing that two hydrogen ions (H+) are released to the soil for every NH + -ion that is nitrified. The implication is that the mineralization of all organic matter (especially urea) acidifies the soil. Where the use of urea or organic matter applications is frequent, maintenance lime applications should be considered for soils with a pH(KCl) lower than 6,0.
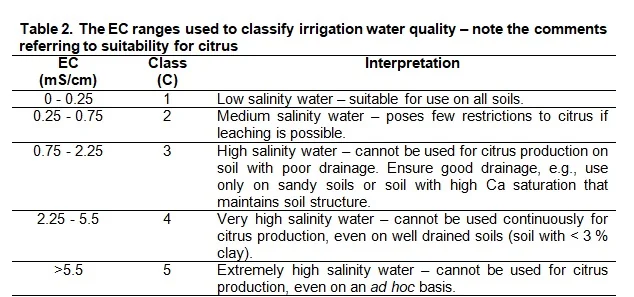
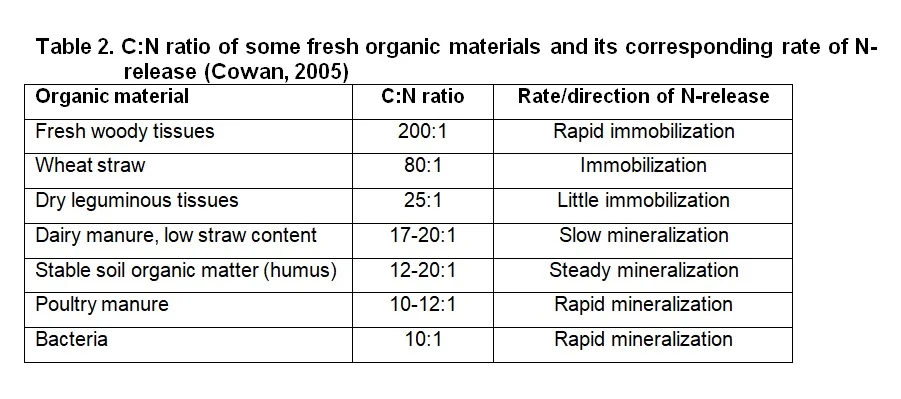
The amount of N, as well as the rate by which it is released, is determined by the C:N ratio of the soil organic matter. The C:N ratio is an expression of the amount of C compared to N that are in the soil, but does not give an indication of the forms that the C and N are in. The value of knowing this ratio is that one can deduce the potential N- availability to both soil micro-organisms and plants. The lower the C:N ratio, the more of the N in the organic matter will be available for mineralization and, therefore, for plant nutrition. On the other hand, a high C:N ratio causes periodic plant N deficiency because the soil micro-organism population is under-supplied by N and as a result consume all the available N, both in the organic and mineral form. This is called immobilisation of mineral N. The addition of organic matter with a high C:N ratio to the vineyard soil can therefore lead to a periodic N deficiency in the vines. In Table 2 an indication of the C:N ratio of some fresh organic materials are given with an indication of its corresponding rate of N-release.
The Rhizosphere
Related to a grape production system, the rhizosphere is the most important facet of soil microbiology. It is the region of soil immediately adjacent to, and directly affected by, the vine roots.
Active growing roots exude organic compounds (exudates) into the soil. These compounds are a food source for micro-organisms, causing their number to be up to 500 times higher than in the rest of the soil (McLaughlin et al., 2000). The characteristics of the exudates changes according to the age of the plant, season, chemical inputs, water availability and nutritional status. The rhizosphere is therefore a constantly changing environment, in which the relative number of each member group adjusts according to the nature of the exudates and the environmental conditions. For example, when a vine wilts due to water stress, more amino acids are released by the roots to the rhizosphere. Likewise, some plants release organic acids into the rihizosphere in response to iron (Fe) or P deficiency, which then solubilise Fe, P, Zn and other metals, thereby enhancing their bioavailability (McLaughlin et al., 2000).
Chemicals also affect the microbial situation of the rihizosphere. It happens in three ways, namely (1) through its direct influence thereon by being incorporated into the soil; (2) by being absorbed by the plant leaves and translocated to other plant parts where it alters the plant's metabolism and, as a result, affects the composition of the exudates; and (3) by being absorbed by the plant, translocated to the roots and exuded by the roots into the rhizosphere. For example, application of a Bordeaux mixture to the leaves of bean plants was found to increase the Cu in the rhizosphere, reducing the bacterial numbers therein (Wilhelm, 1966).
Relating to soil pathogenic infections, the rhizosphere is often called "the first line of defence". Pathogens can only cause a disease by entering the root from the rhizosphere. For it to successfully establish itself as a member of the rhizosphere, it must be able to compete with the other saprophytes for nutrients. But because of the high numbers of microbial organisms and their enhanced activity, which is associated with antagonistic and competing reactions, the rhizopshere generally protects the roots against infection. However, if the balance of this micro-habitat is disturbed by a chemical or detrimental environmental conditions (drought, water-logging, nutritional deficiencies) the effectiveness of the saprophytes to compete with the parasites/pathogens reduces and results in root-infections. In terms of the root environment, biological control of fungal diseases would therefore entail the maintenance of a high activity of beneficial organisms in the root-zone.
Managing Soil Biological Fertility
Although management practices are known to impact the biology of vineyard soils, there is limited knowledge to support the development of detailed management strategies. A particular practice may have the desired result in one situation but have little effect in another because biological communities respond to the interaction of multiple factors including food sources, moisture, historical land use and physical habitat.
A common denominator is that soil potential improves when the number and complexity, or diversity, of the soil biological community increases. There, however, is also an economic limit to the amount of diversity that one might want to obtain. Some practical ideas on increasing and maintaining biological activity and diversity are therefore suggested below.
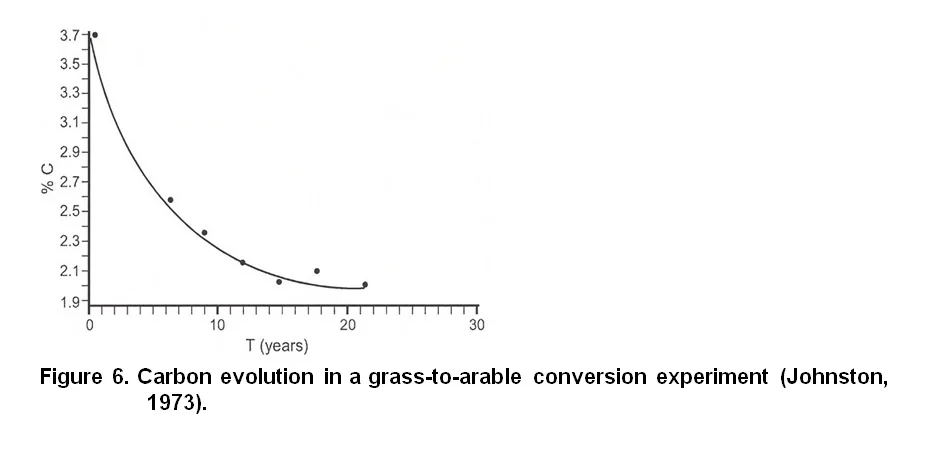
Firstly, supply organic matter to the soil. It is known that over time, the organic matter content of cultivated soils decreases, especially during the first years after development (Figure 6). Organic matter can be added to the soil by utilising crop residue maximally, i.e. do not remove shoots after pruning, by applying compost or manure (if available and affordable) and by planting cover crops. Surface residue, like shoots and stubble, encourages the decomposers, especially fungi because they have an advantage over bacteria in digesting surface residue. Earthworms and arthropods are also benefited. In addition to above ground residue produced, cover crops increase the sub-soil organic matter content through its dense root system. The rhizosphere of cover crop roots also increases the biological activity of the soil significantly. Compost inoculates the soil with a wide variety of organisms while animal manure provides food and mineral nutrients to both larger organisms and micro-organisms. A combination of manure and plant matter will obviously support a larger mixture of soil organisms (Anonymous, 2005).
Secondly, rotate crops. This practice regularly puts a different food source into the soil that results in a wider variety of organisms and prevents the build-up of a single pest species. Within a vineyard context, this can be achieved through rotation of cover crops. For example, on a sandy soil saia oats can be followed up after two years by grazing vetch, which is followed up by pink seradella (personal communication: J. Fourie).
Thirdly, protect the habitat of soil organisms. Soil organisms need air, moisture, a constant food supply and protection against adverse elements. Soil compaction changes the movement of air and water through soil, and may cause a switch from aerobic to anaerobic organisms. It also reduces the space available for larger organisms to move through the soil. The ecosystem is therefore affected, with the result that the population of fungi, larger predators and arthropods declines. Among the nematodes, the predatory species are the most affected, followed by the fungal feeders and then the bacterial feeders. Root-feeding nematodes, however, are the least sensitive to compaction -- perhaps because they do not need to move through the soil in search for food (Anonymous, 2005).
Clean cultivation leads to the migration of most arthropods or to their starvation. The soil climatic conditions are also adverse to any biological activity, while cover crops will stabilize the soil moisture and temperature conditions and maintain the biological habitat. Although tillage enhances bacterial growth in the short term by aerating the soil and mixing the organic matter with the soil, it leads to an accelerated loss of C as CO2 and it triggers explosions of bacterial predators like protozoa. Over the long term the soil organic matter that fuels the organism population reduces and as a result the populations of fungi, earthworms, nematodes and arthropods will decline due to a lack of food, rather than the (also detrimental) mechanical action of tillage.
Pesticides also impacts non-target organisms. It feed some organisms and harms others. Few pesticides have been studied for their effect on a wide range of soil organisms, so that the net effect of moderate chemicals is not well understood. We do know, however, that many of the well-known pesticides reduce soil biological complexity (van Zwieten, 2004). Herbicides often do not affect soil organisms directly, but the weed loss (in clean cultivation) changes the food sources and habitats available to organisms. Fertilisers provide some nutrients to organisms and favour those species that can utilize mineral nutrients. The pH and salt effect of fertilizers like ammonium nitrate and potassium chloride reduces populations of bacteria, fungi and nematodes temporarily (Anonymous, 2005). On the other hand, responsible fertilizer use increases plant growth, and therefore organic inputs into the soil which benefits biological activity. This is especially applicable to cover crops.
In conclusion, it is important to consider the fact that soil biological processes can develop slowly and it can take time before changes are noticed. Some processes can take place at enormous rates, eg. bacterial population growth after soil cultivation, affecting the soil within days, while other processes takes many months or even years to impact the soil.
Practical considerations to keep in mind
The presence of organic matter is a prerequisite for microbial activity. One therefore must ensure its presence. Clean cultivation leads to a loss of soil organic matter, increases soil density and soil erosion, while temperature extremes also develop in the topsoil. All are detrimental conditions to soil biological activity. Rotated cover crop cultivation and the maintenance of pruning material in the vineyard should be an integral part of a viticulturalist's soil management strategy.
The use of organic matter and urea acidifies the soil, regular monitoring of the soil pH.
The C:N ratio of applied organic matter will determine whether N is released rapidly, slowly or whether mineral N in the soil is immobilized periodically. As a general rule, an estimated 50% of all organic N applied to soil will be released during the course of the first year. Thereafter 25% is released in the second year and 10% in the third. Through annual additions of organic material the N released from the soil organic matter will therefore increase continuously. Mineral N-fertilisation will therefore have to be decreased.
The health of the rhizosphere determines to a large extent the health of the vine, particularly its roots system. Many chemicals affect it and as a result, soil conditions that contributes to the rapid restoration of the rihzosphere ecosystem benefits the health and performance of the vine. A high microbial activity in the soil, maintained by the presence of organic matter, is the crucial aspect.
Literature Cited
Anonymous, 2005. Soil biology and soil management. www.extension.unm.edu
Cowan, D. 2005. Building soil organic matter. Agri-Food Laboratories CCA, www.agtest.com/articles/
Dickinson, C.H. & Pugh, G.J.F., 1974. Biology of plant litter decomposition. Academic Press, London, 775pp.
Gregorich E.G., Carter M.R., Doran J.W., Pankhurst C.E. and Dwyer L.M., 1997. Biological attributes of soil quality. In: 'Soil quality for crop production and ecosystem health'. (Eds. E.G. Gregorich, M.R. Carter) pp. 81-113. Elsevier, Amsterdam.
Johnston, A.E., 1973. The effects of ley and arable cropping systems on the amounts of soil organic matter in the Rothamstead and Woburn Ley arable experiments. Rothamstead Experimental Station Annual Report for 1972, 2,131-159.
McLaughlin, M.J., Hamon, R.E., McLaren, R.G., Speir, T.W., & Rogers, S.L., 2000. Review: A bioavailability-based rationale for controlling metal and metalloid contamination of agricultural land in Australia and New Zealand. Aust. J. Soil Res., 38, 1037-1086.
Van Zwieten, L., 2004. Impacts of pesticides on soil biota. In: "Soil Biology in Agriculture." Proc. of a Workshop on Current Research into Soil Biology in Agriculture, (Ed. Lines-Kelly, R.) NSW Dept. of Primary Industries, Tamworth, Australia.
Wilhelm, S., 1966 Chemical treatments and inoculum potential of soil. Annual Review of Phytopathology, 9 (4), 53-78.